The “guardian of the genome”, a title historically given to the p53 protein, evokes immediate curiosity. To be credited as the protector of a cell’s complete set of genetic material is certainly no small feat. Naturally, this leads to a few questions regarding the functions of such a protein, as well as its importance within the cell. In general, the p53 protein and its associated gene, TP53, are best known for their role in protection against cancerous tumors through their control of cellular growth and division. Virtually half of all human cancers involve TP53 or p53 protein mutations, so research into p53’s roles and complexities is critical for cancer protection and prevention in the future. Although p53 has been widely known to function in tumor suppression by inducing cell-cycle arrest or apoptosis, especially under conditions of DNA damage, more recently the established functions, cellular roles, and regulating mechanisms of p53 have expanded immensely.
Tumor suppression by p53: How does it work?
Cells are often faced with numerous stress factors including DNA damage, hypoxia, oncogene activation, and nutrient deficiency, among others. These stresses are the root causes for cancer, abnormal cell growth and mutation that results in malignant tumors. In essence, under normal conditions the p53 protein is in a state of inactivation due to MDM2, an ubiquitin ligase, binding to p53. Upon recognition of tumor-inducing stresses, typically thought to be DNA damage, p53 is activated. It then acts as a transcription factor which induces specific protein-coding or non-protein coding genes such as the cyclin-dependent kinase inhibitor p21. The activated genes then elicit a suitable cellular response such as cell cycle arrest and repair of DNA, senescence, or even apoptosis.
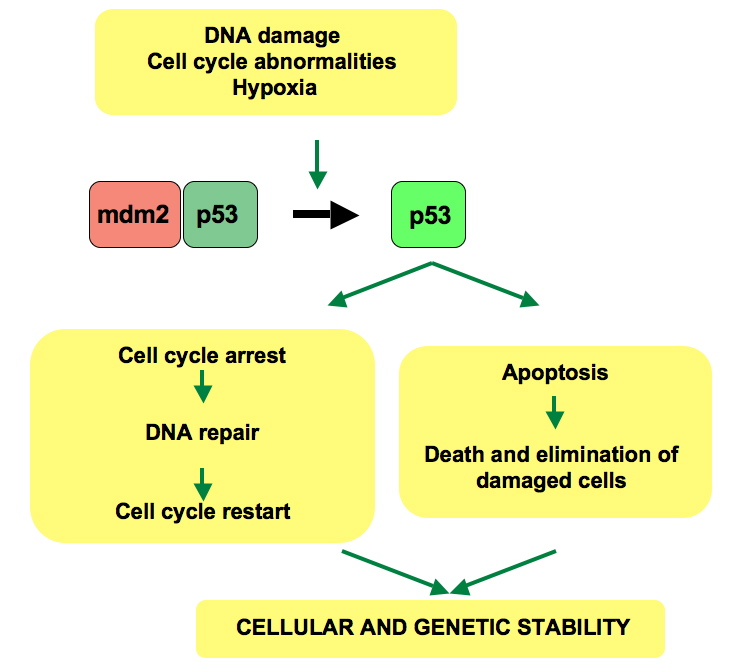
The general pathway for p53’s DNA damage response. Image credit: Thierry Soussi, CC-PD-Mark
So how is p53 regulated?
The disparate cellular responses that can occur due to p53 activity has led to the question: How does p53 determine which response is needed? A number of processes have been proposed, and all likely affect the specific action performed. These regulatory mechanisms include covalent modification of p53, chromatin alteration by p53, as well as the ability for varying quantities of p53 to possess different affinities for p53 response elements (genes). Additionally, p53-interacting proteins such as MDM2 and related proteins play a role in the regulation of p53 activity, and may be affected by feedback loops with p53. The particular response triggered by p53 activity, and thus the cell’s fate, is dependent on the type, extent, and context of the stress present.
The further complexities of p53
As mentioned, p53’s primary function in cancer and tumor prevention has conventionally been thought to be a response to DNA damage, with the end result being cell-cycle arrest or apoptosis, relative to the extent of the damage. However, recently it has been recognized that this p53-induced apoptosis response is not actually the main source of cancer protection provided by p53. Instead, it is more likely p53’s response to oncogenic signalling that is the main source of cancer protection. Oncogenes are genomic sequences that signal uncontrolled cell proliferation. To explain this finding, it is important to note that DNA damage results not only in p53 activated apoptosis, but different forms of cell death as well including mitotic catastrophe. Therefore, DNA damage and its subsequent activation of p53 do not offer additional defense against cancer since cell death occurs independent of p53. Part of the reason p53’s response to DNA damage is so complex is due to the implication of oncogenes during mild DNA damage repair. Typically, mild DNA damage will simply be repaired within the cell. However, in some instances there may be potential oncogenic sequences present, in which case devastating consequences to the cell may occur if these sequences are missed and later transcribed. Therefore, such instances require promotion of senescence, or permanent stoppage of the cell-cycle, by p53 to ensure this does not take place. The question is then raised: Why would p53 promote cell senescence or survival instead of simply initiating cell death on every occasion? It turns out that complete cell death is not always the most sensible response, especially when a cell is exposed to relatively minor stress or damage, and may be able rejoin the healthy cell population. Additionally, the consistent use of apoptosis and cell senescence may greatly reduce the availability of stem and progenitor cells, which results in tissue and organ deterioration, therefore accelerating aging.

Mulitple p53 binding domains associated with DNA. Image credit: Richard Wheeler, CC BY-SA 3.0
Other roles of p53
In addition to tumor suppression, p53 has been discovered to function in a multitude of other roles. The protein p53 has been found to respond to nutritional stresses by promoting autophagy, the digestion of intracellular materials and organelles by lysosomes. Moreover, p53 functions in the regulation of mammalian reproduction by promoting embryo insertion. p53 also influences the cell’s survival by regulating a variety of metabolic pathways including glycolysis, oxidative phosphorylation, and reduction in the output of reactive oxygen species. At first glance this appears to be a paradox, since p53 functions in both the promotion of senescence or cell death, as well as contributing to a cell’s survival. While still not fully understood, the particular role p53 plays is dependent on the type and extent of cell or stress involved, the prevailing gene expression, and the relative activity of both p53 itself and different proteins, among other factors.
Conclusion
The protein p53 is incredibly complex and plays a role in a vast assortment of cellular functions. Classically, p53 has been well attributed to tumor suppression in response to stress, especially that of DNA damage. However, recent research has emphasized the intricacies of p53’s mechanisms and roles within the cell, highlighting key areas that were oversimplified in the past. p53 is involved not only in tumor suppression, but cell survival, reproduction, and numerous other roles. Moreover, the factors that influence and regulate p53’s particular function are wide-ranging. While the specific mechanisms are beyond the scope of this blog, it nonetheless overviews the increasingly understood and multifaceted nature of p53 within the context of the cell, and indeed the organism.
Recent Comments