Why Must a Cell Divide?
Cells are limited in size by their surface areas and volume ratio due to a lack of balanced growth between the parts. Larger cells require more demands on DNA, and it is harder to move nutrients and oxygen across the cell membrane, so they have to divide into two daughter cells. Cell division reduces the cell’s volume which makes it easier to exchange vital materials for cellular processes. It is also important in organisms for reproduction and repair. Then, does cell division always give rise to two identical daughter cells?
The Process of Asymmetric Cell Division
The answer is NO. The mother cell creates two daughter cells with equal cell fates in a symmetric cell division, but two qualitatively distinct daughter cells can be also seen in an asymmetric cell division (ACD). Notably, stem cells can either divide symmetrically or asymmetrically. The asymmetric division occurs when they expand in number during development or after injury. This is more likely used as a tool to maintain appropriate numbers of progeny and control cell fates.
Figure 1. Symmetric versus Asymmetric Cell Division. During a SCD (a), cell fate determining factors are distributed evenly to both arising daughter cells, resulting in identical cell fates. During ACD (b) cell fate determinants segregate unequally into both daughter cells, enabling them to realize different cell fates (Murke et al. 2015).
In principle, asymmetric divisions involve extrinsic or intrinsic factors. With extrinsic factors, daughter cells are initially equivalent, adopting different fates due to the interactions of the daughter cells with each other or with their environment. Intrinsic factors involve different amounts of cell-fate determinants which are distributed into the two daughter cells. The cell biology underlying asymmetric cell division has been studied in various organisms, including a fruit fly, Drosophila melanogaster.
Asymmetric Cell division in Drosophila neuroblast
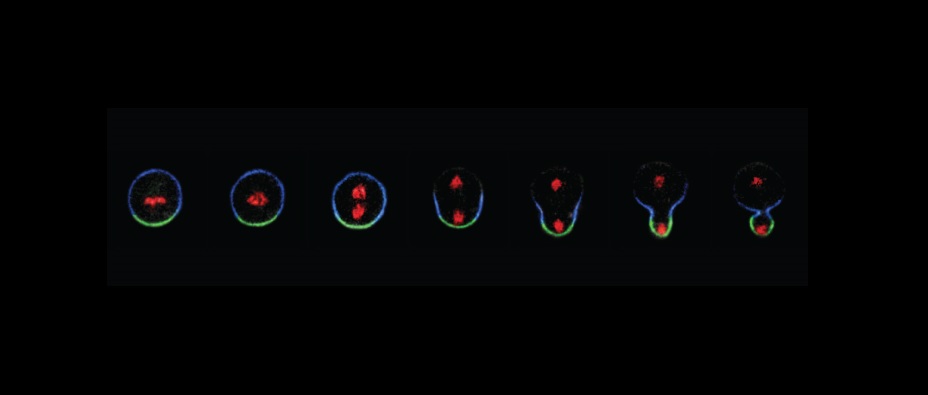
In Drosophila, asymmetric cell division plays an important role in neural development. The cell polarity organizing proteins regulate asymmetric cell division of neuroblast, a neural stem cells. Delamination of the neuroblast from the neuro-ectodermal epithelium in the central nervous system divides asymmetrically along the apical-basal axis, in order to produce a ganglion mother cell (GMC) with a neuroblast. The neuroblast repeatedly undergoes the asymmetric division, while the GMC continues to produce neurons and glia of the central nervous system. The maintenance of apical-basal cell polarity is required for the localization of the cell fate determinants (Prospero and Numb) and the proper orientation of the mitotic spindle. The daughter cell inheriting these two proteins becomes specified, while the second daughter cell with its low levels remains a neuroblast (Fig 2b).
In the peripheral nervous system, each mechanoreceptor (bristles) on the surface of an adult Drosophila, is created by a sensory organ precursor (SOP) cells, which repeatedly undergo asymmetric cell divisions. Figure 2c shows that the SOP divides asymmetrically into pIIa and pIIb daughter cells. The numb is inherited by the pllb cell, which generates the endocytosis and degradation of Notch by removing Notch receptors from the pllb membrane. Therefore, only p11a cell is available for the Notch signaling symmetry pathways. The plla produces hair with a socket cell using the same regulatory mechanisms. The pllb cell produces a glia cell, while the plllb cell further divides to generate the sheath cell and the neuron.
Figure 2. Examples for ACD in Model Organisms. (b) ACD of a Drosophila neuroblast (NB); (c) Drosophila sensory-organ precursor (SOP) development. Localization of cell fate determinants is indicated by crescents (Murke et al. 2015).
Asymmetric cell division and tumor formation
In Drosophila, asymmetric cell division regulates proliferation and self-renewal with cell cycle exit and differentiation in normal stem cells. However, defects in asymmetric cell division turn neural stem cells into tumor-initiating cells, resulting in aberrant self-renewal and impaired differentiation. In other words, tumor formation is from defects in segregating determinants that result in symmetric divisions. Many of the key regulators of asymmetric cell division act as tumor suppressors. Mutations in these tumor suppressor genes would lead to tumor-like over proliferation of neuroblasts. The size of the brains would then become enormous and the animals would eventually die. Tumor suppression can be also followed by transplanting tumor tissue into the abdomen of female host flies, where it continues to grow and kill the host as well.
Figure 3. Asymmetric cell division and tumour formation (Knoblich 2014).
The identification of key regulators of asymmetric cell division (Lgl and Dlg) and a segregating determinant (Brat) suggests that tumors arise from defects in asymmetric cell division. The transplantable tumors also form in mitotic neuroblast clones mutant for Numb and Prospero.
Lastly, identification of the relationship between asymmetric cell division and cellular immortalization can be further studied since defects in asymmetric cell division are relevant for human tumorigenesis and this could be part of the mechanisms that convert a normal mammalian stem cell into a cancer stem cell.



Recent Comments