Introduction
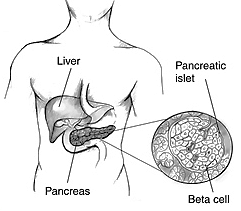
In a typical individual, after a meal or when blood glucose levels spike, pancreatic islet beta cells are stimulated to produce the hormone insulin. Insulin then binds to receptors of various cells in the body to allow for absorption, storage, usage, and regulation of blood glucose. However, in patients with type I diabetes pancreatic islet cells have been destroyed by the immune system leading to a limitation in absorption, usage and regulation of blood glucose. As a result, patients must revert to artificial insulin injections to control their blood glucose levels. Unfortunately, over years of artificial regulation of diabetes, comorbid complications become increasingly prevalent such as, heart disease, kidney disease, and nerve or eye damage.
Figure 1.0 Illustration of Beta Pancreatic Cells.
Incident cases of Type I diabetes mainly occur in children of northern European descent. As type I diabetes is more prevalent among certain ethnic groups it would be reasonable to think that its aetiology is solely due to genetics, however this is not the case. The development of type I diabetes involves an exceedingly complex relationship between genetics, exposure to vitamin D and sunlight, breastfeeding, infantile obesity, viral infections, and many others. Due to these multifactorial complexities, teasing apart the many plausible causes of type I diabetes is exceedingly difficult. Nonetheless, in Canada we have about 27,000 children under the age of 19 alone, living with type I diabetes.
Solution!
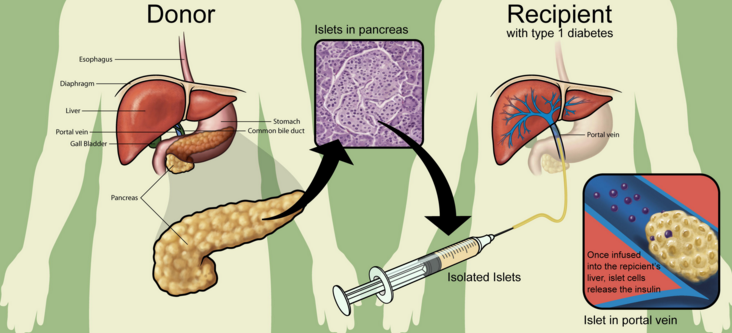
Pancreatic islet transplantation is a procedure in which islets from the pancreas of a deceased organ donor are purified, processed, and transferred into another person. This Procedure is termed the Edmonton Protocol and is an allo-transplant, as cells are taken from a genetically dissimilar individual. Once implanted, the new islets begin to make and release insulin. The goal of islet transplantation is to infuse enough islets to control the blood glucose level eliminating the need for insulin injections and pumps. Since islet cells make up only 1-2% of the pancreas, it seems more feasible to focus on the transplantation or infusion of these cells rather than the whole pancreas. In addition, transplanting a whole pancreas carries a higher risk of mortality during the surgical procedure, as opposed to islet cell infusion.
What Does the Procedure Entail?
A typical islet cell transplant requires about 1 million islets, which is equal to two donor pancreatic organs. In order for islet cells to be viable for the recipient, transplantation must occur immediately.
The transplant procedure itself is easy and takes less than an hour to complete and does not involve as much of a surgical risk as a full pancreas transplant. During the procedure the surgeon uses an ultrasound or x-ray imaging to guide the placement of a small catheter through the abdomen and into the liver. The islet cells are then injected through the catheter into the liver. Once in the liver, it takes some time for the cells to attach to new blood vessels and for them to begin generating insulin. After the transplant procedure, glucose levels will be monitored, and further insulin injections may be needed until self-control/regulation by the new cells is achieved.
Figure 2.0 Depiction of the Edmonton Protocol.
Benefits
A successful transplant may reduce co-morbidities associated with type I diabetes through more efficient regulation of blood glucose. Moreover, transplanting islet cells has clear advantages over full pancreatic transplants. Firstly, unlike a pancreatic transplant, islet cell transplantation is a minor surgical procedure, less expensive, and much safer. Secondly, scientists and medical clinicians are in the process of learning how to protect beta cells from an attack by the immune system. Once successful, in finding the exact mechanisms through which the autoimmune attack occurs, patients with already transplanted islet cells may have a chance of becoming independent of immunosuppressant drugs.
Future Studies
Continuing research in this area is necessary to improve procedures of transplant therapy as well as surgical techniques. The use of animal islet cells for humans seems to be more viable, as the supply of human islets is severely limited. Perhaps scientists may find a way to extract and use animal islet cells for human use, similar to how insulin was initially mass produced from swine pancreatic cells for human use. With the existing sources of human islet cells, even the current most successful method of human islet transplantation could only ‘cure’ a small fraction of the population living with insulin dependent diabetes. If islets from animal sources (for example, pigs) could be made to work successfully, a cure would be available for everyone. But transplants between species involve a whole new set of problems when it comes to regulating the body’s immune response, so much work will still has to be done to make that a possibility. In addition, future studies will be focusing on making transplantable islets more abundant through the use of adult or embryonic stem cells.
- All images permitted for reuse as filtered by Google images

Recent Comments