Designer Babies
Human embryos that have been genetically selected or modified, either to ensure the absence of a defective gene that will render them nonviable/abnormal, or to have a specific gene (such as genes that codes for blonde hair and blue eyes) present, are sometimes called “designer babies.” In the past, the purpose of designer babies was to provide stem cells for an older, diseased sibling, which was the case in the first recorded designer baby event in 2000. Since then, this practice has continued; however, with modern gene editing technology, science decided to take designer babies one giant step forward. With the use of CRISPR/Cas9, scientists hope to be able to edit the genome of human embryos with a high degree of precision, and thus allow for ultimate customizability of the genome. This method of genome modifications in embryos would provide prenatal therapy for genetic diseases, such as cystic fibrosis, Duchenne muscular dystrophy, and Alzheimer’s. Conversely, the same methodology could be used to modify traits in the newly formed embryo. These traits include, but are not limited to, physical characteristics, mental acuity, and athletic potential. This provides basis for intensive ethical debate, however this is not ethics class! From a purely scientific standpoint, the concept of designer babies via modification by CRISPR is simply awesome.

Figure 1. Retrieved from https://prezi.com/c_dy03ykvihu/designer-babies/, photo credit to Janel DiBiccari
CRISPR/Cas9
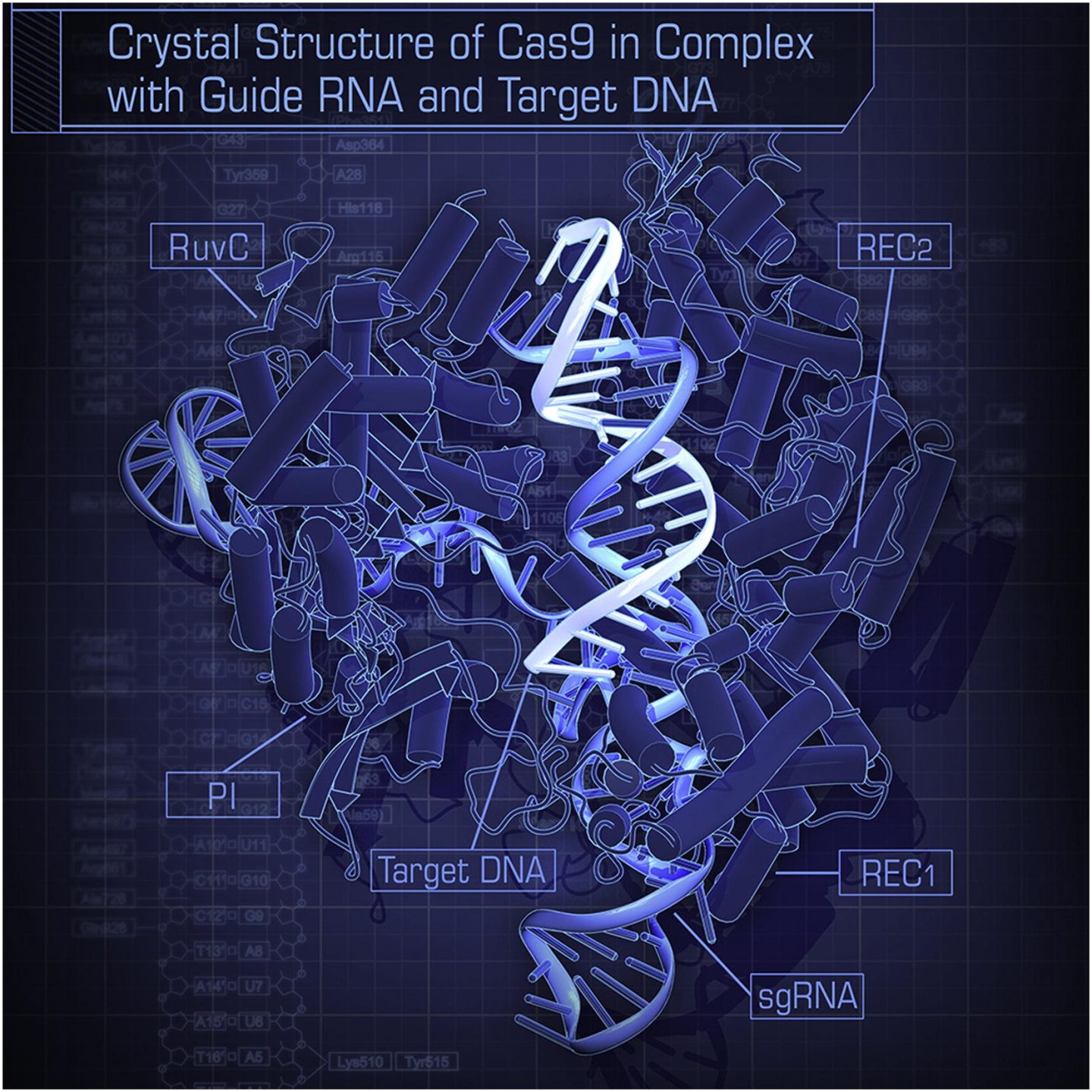
Clustered Regularly Interspaced Short Palindromic Repeats, more easily referred to as CRISPR, is today’s premier tool in genetic engineering. It was originally developed to “knock-out” certain genes, however now it has been modified to activate or repress expression of certain genes, as well as edit genes entirely by adding or deleting nucleotides. The CRISPR/Cas9 system requires the non-specific CRISPR associated endonuclease 9 (Cas9) as well as a guide RNA (gRNA). The purpose of Cas9 is to read and edit the DNA, while the purpose of the gRNA is to recognize areas of the genome homologous to its own sequence. The gRNA is a synthetic strand of RNA produced by the genome engineers to target their gene of interest. The specificity of the gRNA for the gene of interest proves to be the major player in CRISPRs specificity to the target gene; it must be specific enough to locate the target gene and no other genes in the genome. The specificity of the gRNA proves to be the prominent restriction with CRISPRs current low precision. If the gRNA exhibits any specificity to another spot on the genome, then the complex will bind and edit the DNA in unintended positions.
Human Trials
Many believe that this technology is somewhat science fiction, or at least not feasible for use in the near future. However, work on editing human embryos has already taken place; in 2015, researchers in China published the first research of its kind on human embryos. They took 86 non-viable embryos from fertility clinics that had mutations in their human β-globin genes that could cause a fatal blood disorder and attempted to render them viable. They started with single celled zygotes, and after using the CRISPR technique with the proper gRNA, allowed them to develop to the “8 cell stage.” Some did not survive, however in the ones that did, it was found that a small fraction actually underwent successful repairs. Most of them also had modifications done to other parts of their genomes, illustrating the current inaccuracy of CRISPR. Some researchers attributed these mutations to the fact that these were not healthy, normal embryos. This hypothesis may very well hold true, and now it can be tested. Recently, the Human Fertilization and Embryology Authority in the UK has given developmental biologists permission to experiment on healthy human embryos using CRISPR/Cas9. The main scope of their study is to suppress the gene OCT4, which is active in cells that continue development into the fetus, and let normal development continue for one week until the blastoderm stage. This is exciting, because now the techniques surrounding gene editing may be able to be perfected, providing vast biomedical applications; this could be the beginning of the treatment of genetic diseases before the embryo develops.
Embryonic Development and Implications
Development in an embryo, following CRISPR modification, is unchanged and will continue normal development. However, if an embryo were to be modified and then birthed, there is great debate surrounding the outcomes of the modified individual’s offspring. Since modification is done at the single cell zygote stage, such modifications are known as germline modifications, because subsequent cell divisions will form germ cells at some point. These modifications being inherited have unknown effects on future generations because of the fact that it is not simply just modifying a single gene that will produce a character trait, but how genes interact with one another. It is thought that DNA modification to the germ line of an organism will not affect normal development of its progeny, however will increase tumour incidence later in its adult life.
The CRISPY Conclusion
There are many possibilities surrounding designer babies, ranging from disease treatment to the modification of traits. There has been much speculation in the scientific community about modifying human embryos; will people exploit this technology to create the “perfect,” biologically superior human being? Will human embryo modification change society and the human race? This sounds like a great movie idea. For now, the technique (CRISPR/Cas9) that could possibly induce genetic modifications is severely inadequate, and will require much more research and improvement before it could even be thought of being used in practice. However, with advancements in the CRISPR/Cas9 technique, we could potentially introduce a generation free of the genetic diseases that once plagued our society.
Recent Comments